New pre-print: VHL inhibitor binding increases intracellular level of VHL
Great work by Julianty, in collaboration with Sonia Rocha
Authors: Julianty Frost, Sonia Rocha*, Alessio Ciulli*
Title: VHL inhibitor binding increases intracellular level of VHL
Abstract
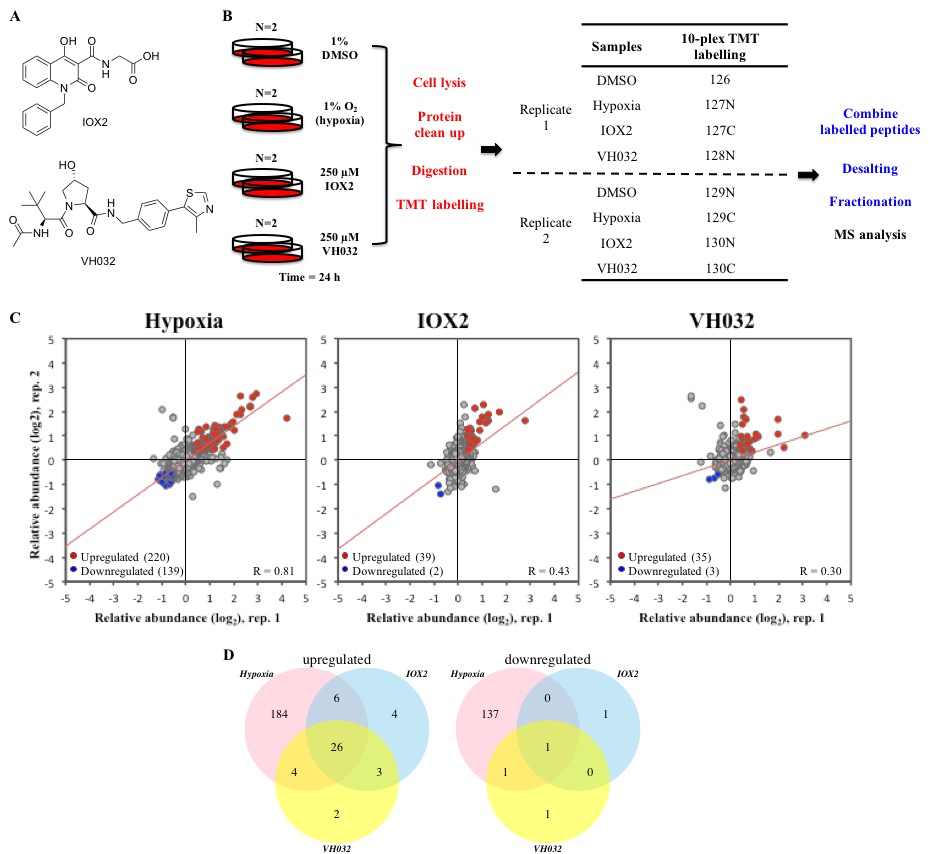
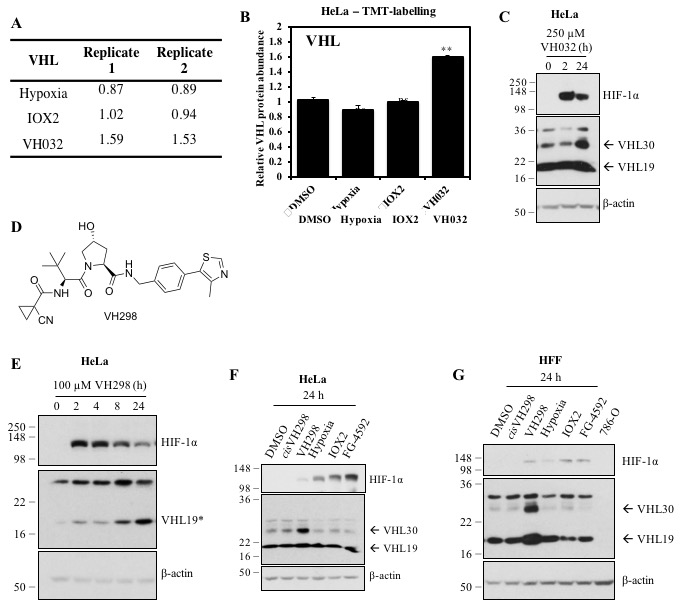
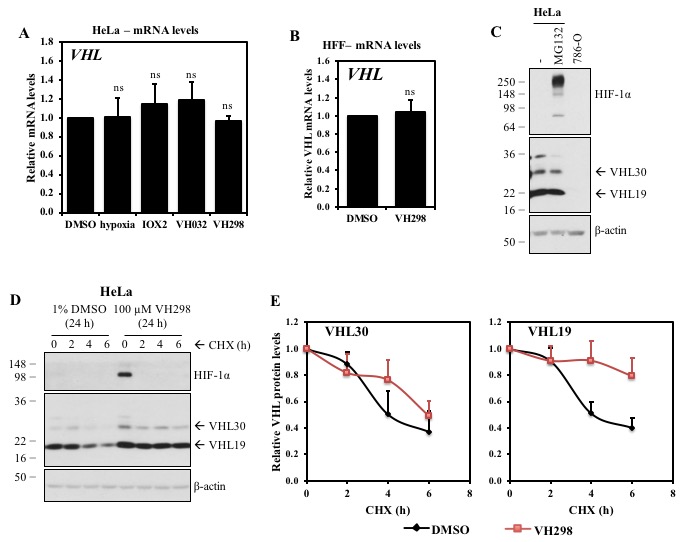
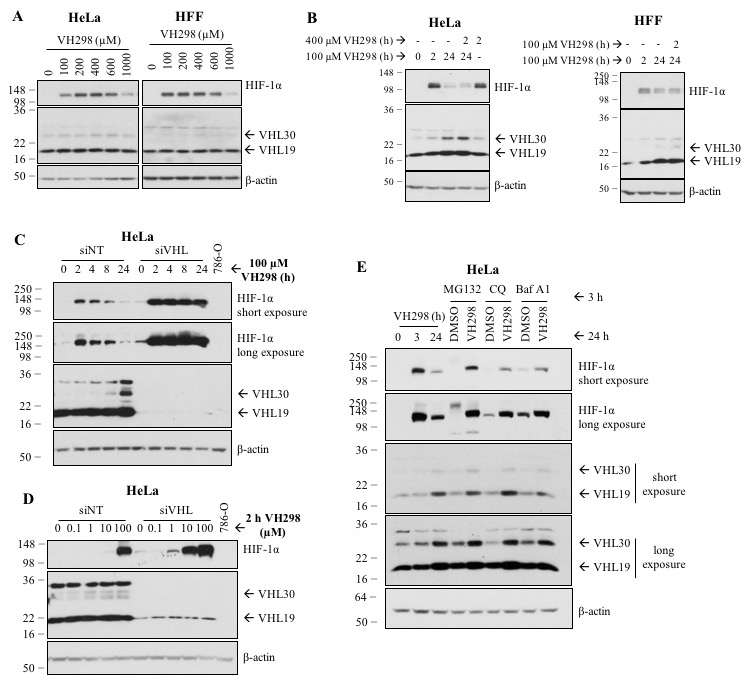
The von Hippel-Lindau (VHL) protein is a tumour suppressor protein frequently mutated in the VHL disease, which functions as substrate recognition subunit of a Cul2 E3 ubiquitin ligase (CRL2VHL). CRL2VHL plays an important role in oxygen sensing, by binding and targeting Hypoxia Inducible Factor-alpha subunits (HIF-alpha) for ubiquitination and degradation. VHL is also commonly hijacked by heterobifunctional degrader molecules known as proteolysis-targeting chimeras (PROTACs). In previous work we reported the structure-based design and functional characterisation of VHL inhibitors (VH032 and VH298) that induce the HIF response in cells. Here, we use unbiased quantitative mass spectrometry to identify the proteomic changes elicited by the VHL inhibitor and compare this to hypoxia or broad-spectrum prolyl-hydroxylase domain (PHD) enzyme inhibitor IOX2. Our results demonstrate the VHL inhibitor selectively activates the HIF response that vastly overlaps with hypoxia- and IOX2-induced proteomic changes. Interestingly, VHL inhibitors were found to selectively upregulate a single protein, which is VHL itself. Our analysis revealed that this occurs via protein stabilisation of VHL isoforms and not via changes in transcript levels. Increased VHL levels upon VH298 treatment resulted in turn to reduced levels of HIF-1 protein. Our results demonstrate the high specificity of VHL inhibitors and suggest that use of these inhibitors would not produce overtly side effects due to prolonged HIF stabilisation. They also exemplify the concept that small-molecule binding induced protein stabilisation can increase protein levels inside cells.
Update (23 June 2021): Full paper now published in J. Biol. Chem.