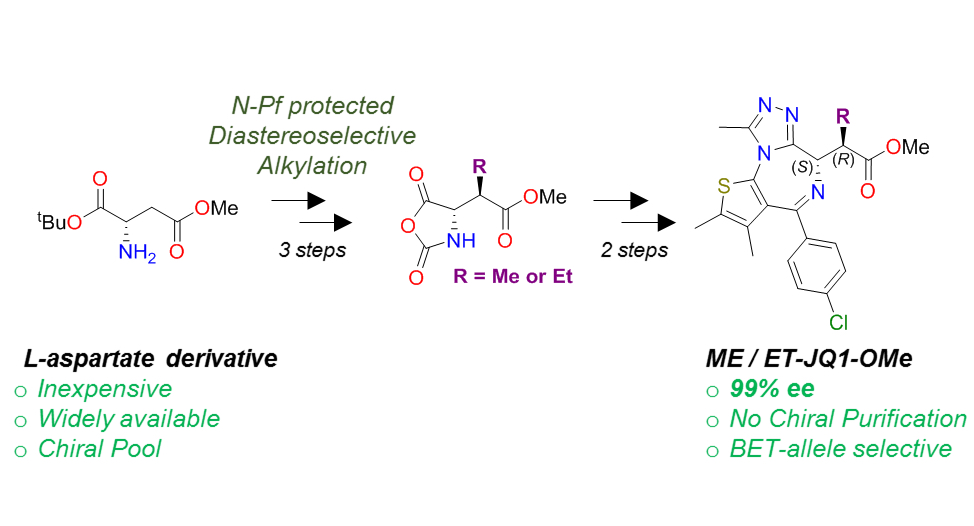
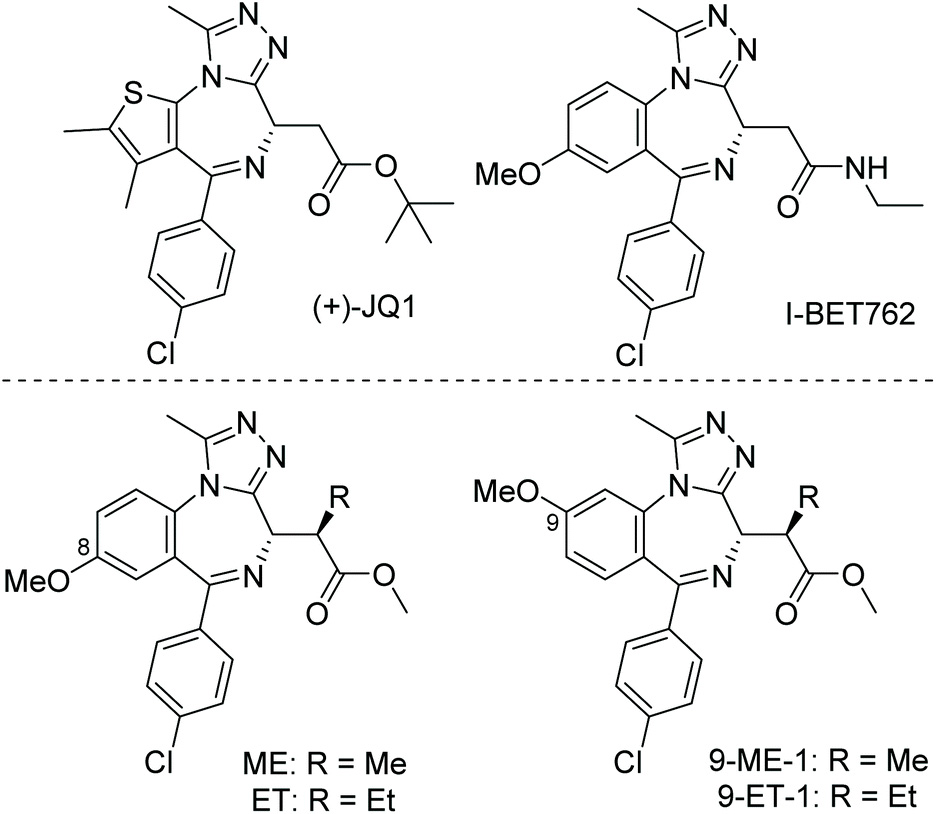
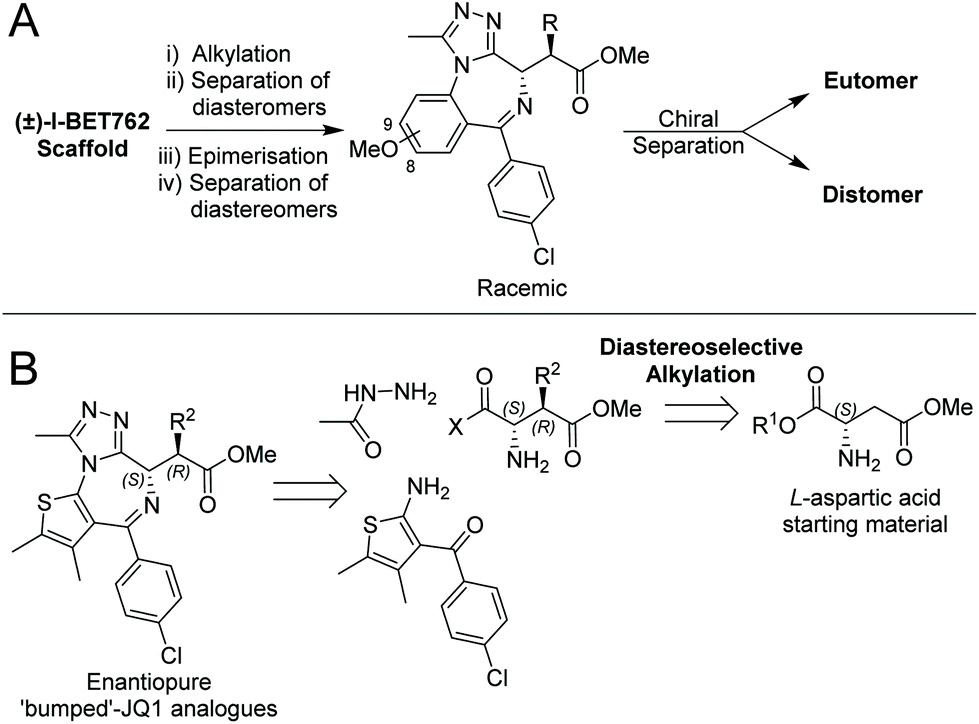
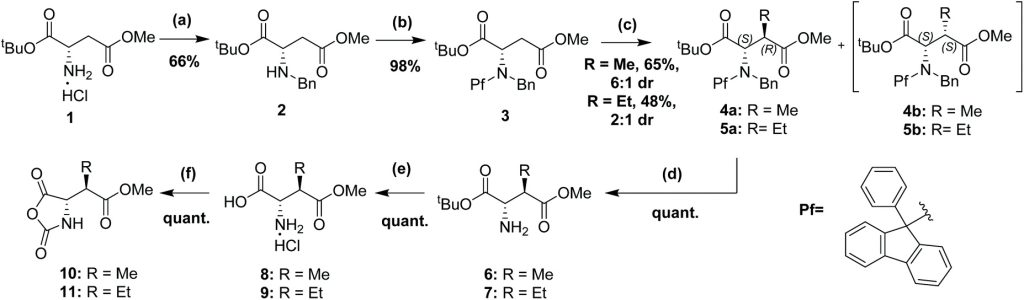
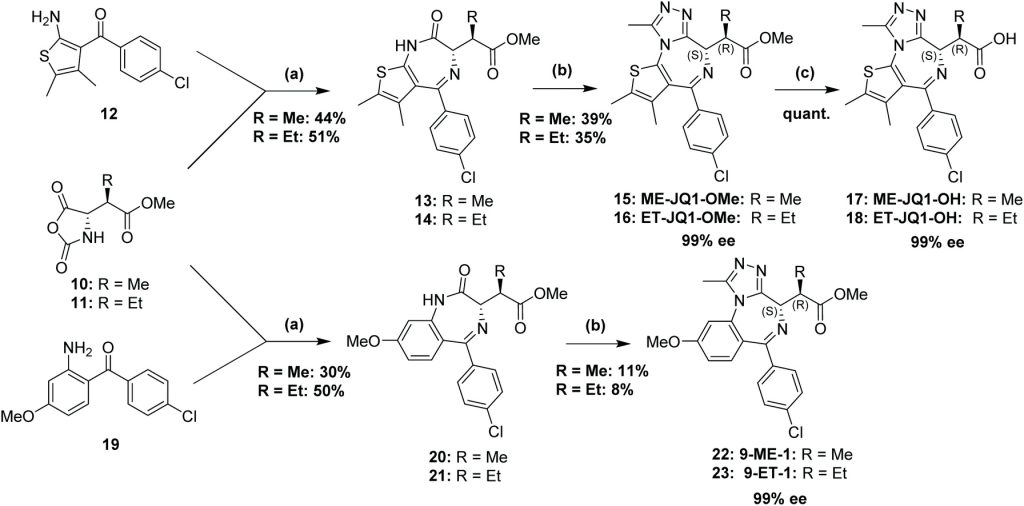
Our new paper describing stereoselective syntheses of allele-selective BET inhibitors bumped-JQ1 is now out in the RSC journal Org. Biomol. Chem.
Congratulations to Adam and Andrea, on their development of this new route to synthesise the compounds!
Read the Open Access full article.
Authors: Adam G. Bond, Andrea Testa and Alessio Ciulli*
Title: Stereoselective synthesis of allele-specific BET inhibitors
Abstract
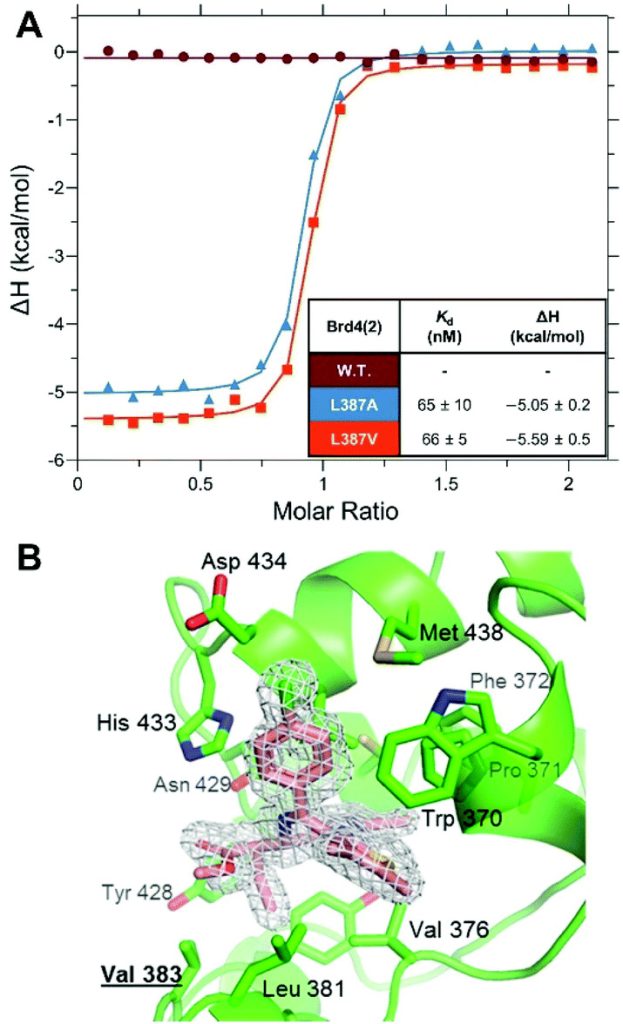
Developing stereoselective synthetic routes that are efficient and cost-effective allows easy access to biologically active molecules. Our previous syntheses of allele-selective bumped inhibitors of the Bromo and Extra-Terminal (BET) domain proteins, Brd2, Brd3, Brd4 and BrdT, required a wasteful, late-stage alkylation step and expensive chiral separation. To circumvent these limitations, we developed a route based on stereocontrolled alkylation of an N-Pf protected aspartic acid derivative that was used in a divergent, racemisation-free protocol to yield structurally diverse and enantiopure triazolodiazepines. With this approach, we synthesized bumped thienodiazepine-based BET inhibitor, ET-JQ1-OMe, in five steps and 99% ee without the need for chiral chromatography. Exquisite selectivity of ET-JQ1-OMe for Leu-Ala and Leu-Val mutants over wild-type bromodomain was established by isothermal titration calorimetry and X-ray crystallography. Our new approach provides unambiguous chemical evidence for the absolute stereochemistry of the active, allele-specific BET inhibitors and a viable route that will open wider access to this compound class.